In the field of weight management therapies, an emerging drug is Semaglutide. It is a GLP-1 receptor agonist originally approved for treating type 2 diabetes (T2D). Semaglutide weight loss therapy has recently made headlines.
With intriguing evidence pointing to significant weight loss potential, researchers are focusing on the potential of Semaglutide. As a GLP-1 receptor agonist, Semaglutide functions in a unique way. It affects appetite regulation & energy balance.
This article explores Semaglutide weight loss therapy, its emerging role, and its potential impact on the field.
As we dive into the details, we will consider the following:
- Semaglutide mechanism of action and its implications for weight loss,
- The current state of research on semaglutide as a weight loss treatment,
- Future directions for semaglutide therapy in weight
So if you are interested in researching Semaglutide, this guide offers detailed insights into this GLP-1 receptor agonist. Let’s start our journey.
Part 1. What is Semaglutide?
Semaglutide is an innovative and promising treatment that operates as a glucagon-like peptide-1 (GLP-1) receptor agonist. It belongs to the GLP-1 family, renowned for stimulating insulin and decreasing glucagon secretion. Semaglutide shares a 94% homology to human GLP-1. So it ensures a reduced risk of immunogenicity. (Kalra et al., 2020)
Structure and Unique Characteristics of Semaglutide
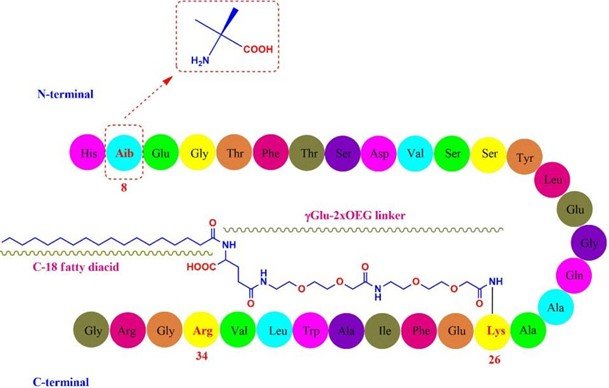
Chemically, Semaglutide presents as a peptide composed of 31 amino acids. This peptidic structure features certain unique attributes. Here’s an overview of these attributes:
- Semaglutide has an alanine residue at the 8th position, which is substituted with 2-aminoisobutyric acid. This particular substitution safeguards semaglutide from degradation by the DPP-4
- The peptide also contains an arginine substitution at the 34th Originally a lysine residue in native GLP-1 is present here. This change facilitates the production of GLP-1 analogs through a semi-recombinant process.
- Finally, a lysine residue at the 26th position has been acylated to allow for the attachment of a C18 fatty A hydrophilic linker, “γGlu-2xOEG”, is used for this attachment. This addition elongates the systemic half-life through enhanced albumin binding and reduced renal clearance. (Al Musaimi et al. 2018)
Enhanced Stability and Prolonged Half-Life of Semaglutide
One of the key highlights of semaglutide is its superior stability and prolonged half-life. It distinguishes this peptide from other GLP-1 receptor agonists.
The peptide is modified to increase the affinity of the pending fatty acid moiety for albumin. This modification increases the stability and half-life. Furthermore, the unique composition enables semaglutide to resist enzymatic degradation.
So Semaglutide peptide has the potential for once-weekly administration. All these changes have also improved its usability as a potential treatment for type 2 diabetes.
The pharmacokinetic studies conducted on the Göttingen mini pig model, which simulates moderate insulin deficiency and diabetes, along with in vivo efficacy in anti-hyperglycemic and body weight lowering effect on obese, hyperinsulinemic db/db mice model (for type 2 diabetes), propelled semaglutide to be chosen as a promising clinical trial candidate. (Mahapatra et al. 2022)
You can legally buy Semaglutide online from NuScience Peptides here!
Now let’s discuss the Semaglutide mechanism of action and its effects.
Part 2. Semaglutide Weight Loss Mechanism of Action & Effects
Semaglutide exerts its effects by activating GLP-1 receptors. Glucagon-like peptide-1 is a major incretin hormone in humans. It is recognised for its multiple roles in metabolism and glucose regulation.
Through activating these receptors, semaglutide augments insulin secretion in a glucose-dependent manner. It also inhibits glucagon release and suppresses hepatic
gluconeogenesis. On top of that, GLP-1 receptor activation by semaglutide helps delay gastric emptying. Ultimately, Semaglutide weight loss occurs by reducing appetite and energy intake.
Let’s take a detailed look at the effects of Semaglutide.
Semaglutide’s Effects on Various Body Systems
Semaglutide impacts multiple body systems:
● Pancreas
By stimulating the GLP-1 receptors in the pancreas, semaglutide enhances insulin secretion. This aids in maintaining proper blood glucose balance.
● Gastrointestinal Tract
Semaglutide slows gastric emptying. It leads to a feeling of fullness and a reduction in overall food intake.
● Fat Tissue
It is understood that GLP-1 receptor agonists can also influence fat tissue by reducing lipolysis. It can contribute to overall weight loss.
● Central Nervous System
Semaglutide interacts with GLP-1 receptors in the brain, particularly in appetite & reward-related regions. Activation of these receptors leads to decreased responses to food stimuli.. (Mahapatra et al. 2022)
Out of all these effects, here’s a very significant outcome of Semaglutide.
Semaglutide, Appetite Suppression, and Satiety Induction
One of the significant outcomes of semaglutide’s action in the body is its effect on appetite and satiety.
The delayed gastric emptying caused by semaglutide leads to an extended feeling of fullness. Furthermore, stimulating GLP-1 receptors in the brain influences the neural response to food. It leads to reduced cravings and decreased food intake.
Evidence of semaglutide’s role in appetite suppression & satiety induction was observed in a study. It was a randomised, crossover, placebo-controlled trial. The study involved obese type 2 diabetes patients and normoglycemic obese and lean individuals.
The trial found that obese subjects and patients with type 2 diabetes showed:
- Increased brain responses to food pictures in appetite and reward-related brain (Ard et al. 2021)
The Result: Semaglutide Weight Loss
By acting in the manner described above, semaglutide effectively induces weight loss. The combined effects of:
- Insulin regulation,
- Decreased glucagon release,
- Slowed gastric emptying,
- Appetite suppression,
contribute to reduced body weight. Moreover, the unique design of semaglutide enables it to resist proteolytic degradation. It allows Semaglutide to function longer in the body and provides sustained benefits for individuals managing their weight.
Now let’s look at some clinical studies on Semaglutide weight loss therapy.
Part 3. Semaglutide Weight Loss Clinical Studies
Numerous clinical trials have explored its efficacy and safety. They revealed significant potential in facilitating weight loss and improving metabolic health. This body of research presents compelling evidence for using semaglutide in the fight against the global obesity epidemic.
The following sections will delve into the details of four pivotal studies. These studies shed light on the multifaceted benefits of semaglutide.
Study 1: Wider Benefits of Semaglutide in Obesity from the STEP Program
This study evaluated the impact of semaglutide on quality of life, control of eating, and body composition in adults with obesity. The results of the study showed that:
- Semaglutide significantly improved all IWQOL-Lite-CT scores related to physical function and psychosocial
- Improvements in all Control of Eating questionnaire domains were noted up to week There were significant differences in craving control and craving for savoury lasting until week 104.
- The total fat mass reduction was greater with semaglutide than with (O’Neil et al., 2022)
Study 2: Weekly Semaglutide Use in Adults with Overweight or Obesity
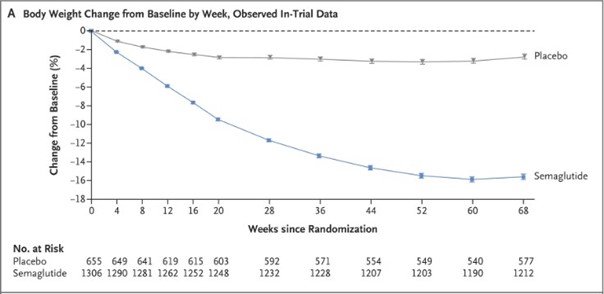
This trial examined the effects of semaglutide on body weight, cardiometabolic risk factors, physical functioning, and adverse events. The results are:
- Participants on semaglutide had an average weight loss of 9% at week 68 compared to a 2.4% loss in the placebo group.
- More participants in the semaglutide group achieved weight reductions of 5% or more (86.4% vs 5%), 10% or more (69.1% vs 12.0%), and 15% or more (50.5% vs 4.9%).
- Improvements were seen in cardiometabolic risk factors and physical functioning in the semaglutide
- Nausea and diarrhea were common but generally mild and transient. Gastrointestinal events led to more treatment discontinuations in the semaglutide group (4.5% vs 8%). (Wilding et al. 2021)
Study 3: Semaglutide’s Impact on Energy Intake, Appetite, and Gastric Emptying
This study investigated semaglutide’s effects on energy intake, appetite, control of eating, gastric emptying, and safety. Here are the results:
- Semaglutide reduced energy intake by 35% compared to
- Significant reductions in hunger and prospective food The increases in fullness and satiety were also reported in the semaglutide group.
- Better control of eating and fewer/less intense food cravings were reported with
- No evidence of delayed gastric emptying was The safety was consistent with the known profile of semaglutide. (Friedrichsen et al. 2021)
Study 4: Semaglutide as a GLP-1 Receptor Agonist for Type 2 Diabetes Management
This review focused on semaglutide’s development, clinical studies, pharmacotherapy, and more.
- Semaglutide demonstrated strong anti-hyperglycemic activity in multiple clinical trials involving adults, elderly & obese type 2 diabetic
- Despite gastrointestinal side effects, semaglutide was well-tolerated. It provided better glycemic control with a low risk of hypoglycemia and good patient
- The SUSTAIN-6 and PIONEER-6 studies confirmed the cardiovascular safety of semaglutide for long-term (Mahapatra et al. 2022)
Now let’s move on to the dosage and administration of Semaglutide.
Part 4. Semaglutide Dosage & Administration
Semaglutide is typically administered in a controlled & gradually escalating dosage. It maximises its weight loss benefits and minimises potential side effects. This stepwise approach begins at a low dose and progressively increases over time. The recommended starting dose is:
- 25mg weekly for the first four weeks.
- The dosage is then increased to 5mg in weeks 5-8.
- Increased to 1mg in weeks 9-12.
- Then 7mg is given in weeks 13-16.
- From week 17 and onwards, the full dose of 4mg per week is administered.
This medication is administered once a week via subcutaneous injection. The duration of treatment in clinical studies ranges from 52 to 104 weeks. It depends on the objectives and parameters of the study. It’s also critical to note that the dosage should not exceed 2.4mg per week.
Adherence to this dosing protocol is crucial for the optimal effectiveness of semaglutide. In the event of a missed dose, it can be taken within five days. However, the dose should be skipped entirely if this window is missed. (Chao et al., 2022) (Alabduljabbar et al. 2022)
After looking at the dosage, here’s some information on the safety profile of Semaglutide weight loss therapy.
Part 5. Semaglutide Weight Loss Safety Profile
Semaglutide has met stringent safety standards set by regulatory authorities like the FDA. Its safety profile was established pre- and post-marketing to identify and manage potential rare adverse events.
Here are some effects shown by a study.
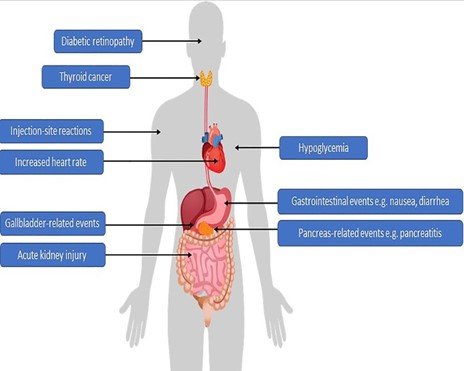
Common Side Effects and Adverse Events
- Some side effects of GLP-1RAs include acute kidney injury, pancreatitis, pancreatic cancer, and thyroid However, evidence has largely dismissed these concerns about Semaglutide.
- Increased gallstones have been associated with Semaglutide
Specific Precautions
- The co-product in oral Semaglutide, SNAC, can be toxic at high But the dosage in each tablet of Semaglutide is well below the toxic threshold.
- Ongoing post-marketing surveillance will identify any real-world safety profile differences between the subcutaneous and oral forms of
Application to Clinical Practice
Safety data on Semaglutide is mainly from controlled clinical trials, sometimes limiting generalizability. Real-world evidence hasn’t shown significant discrepancies in the incidence of side effects like hypoglycemia or drug discontinuation rates.
So continuous surveillance and research are crucial to ensure Semaglutide’s continued safe use. (Smits et al. 2021)
If you want to buy Semaglutide for research purposes, just visit NuScience for some quality products.
Part 6. Buy Semaglutide Online from NuScience Peptides
For those seeking to buy Semaglutide online for research purposes, NuScience Peptides offers a reliable and high-quality service. They prioritise customer satisfaction, offering:
● Same Day Shipping
All orders of in-stock items placed before 12:00 pm EST are shipped the same day, ensuring swift delivery.
● Dedicated Service
NuScience Peptides is committed to superior customer service, ready to assist with any queries or concerns.
● Free Shipping
To enhance customer experience, free shipping is offered on all orders totalling $125 or more.
● Purist Quality
Ensuring high product quality is at the core of NuScience Peptides’ ethos. All products undergo rigorous lab testing and certification for maximum purity.
The NuScience Peptides community comprises many researchers and scientists. They rely on the company’s quality, precision, and expertise for their critical projects. By choosing NuScience Peptides, you align with a trusted ally in advancing scientific understanding.
 Semaglutide Peptide From NuScience Peptides
Semaglutide Peptide From NuScience Peptides
Conclusion
Semaglutide weight loss therapy showcases exciting potential. Its effectiveness in managing chronic overweight and obesity conditions makes it a promising candidate for further research. We must stay informed and prepared as we anticipate new developments in Semaglutide weight loss therapy. For researchers aiming to explore the potential of Semaglutide further, NuScience Peptides offers a reliable, high-quality source.
Reference
Al Musaimi, O., Al Shaer, D., de la Torre, B. G., & Albericio, F. (2018). 2017 FDA peptide harvest. Pharmaceuticals (Basel, Switzerland), 11(2), 42. https://doi.org/10.3390/ph11020042
Alabduljabbar, K., Al-Najim, W., & Le Roux, C. W. (2022). The Impact Once-Weekly Semaglutide 2.4 mg Will Have on Clinical Practice: A Focus on the STEP Trials.
Nutrients, 14(11), 2217. https://doi.org/10.3390/nu14112217
Ard, J., Fitch, A., Fruh, S., & Herman, L. (2021). Weight loss and maintenance related to the mechanism of action of glucagon-like peptide 1 receptor agonists. Advances in Therapy, 38(6), 2821–2839. https://doi.org/10.1007/s12325-021-01710-0
Chao, A. M., Tronieri, J. S., Amaro, A., & Wadden, T. A. (2022). Clinical insight on semaglutide for chronic weight management in adults: Patient selection and special considerations. Drug Design, Development and Therapy, Volume 16, 4449–4461. https://doi.org/10.2147/dddt.s365416
Friedrichsen, M., Breitschaft, A., Tadayon, S., Wizert, A., & Skovgaard, D. (2021). The effect of semaglutide 2.4 mg once weekly on energy intake, appetite, control of eating, and gastric emptying in adults with obesity. Diabetes, Obesity & Metabolism, 23(3), 754–762. https://doi.org/10.1111/dom.14280
Kalra, S., & Sahay, R. (2020). A review on semaglutide: An oral glucagon-like peptide 1 receptor agonist in managing type 2 diabetes mellitus. Diabetes Therapy : Research, Treatment and Education of Diabetes and Related Disorders, 11(9), 1965–1982. https://doi.org/10.1007/s13300-020-00894-y
Mahapatra, M. K., Karuppasamy, M., & Sahoo, B. M. (2022). Semaglutide is a glucagon-like peptide-1 receptor agonist with cardiovascular benefits for managing type 2 diabetes. Reviews in Endocrine & Metabolic Disorders, 23(3), 521–539. https://doi.org/10.1007/s11154-021-09699-1
O’Neil, P. M., & Rubino, D. M. (2022). Exploring the wider benefits of semaglutide treatment in obesity: Insight from the STEP program. Postgraduate Medicine, 134(sup1), 28–36. https://doi.org/10.1080/00325481.2022.2150006
Smits, M. M., & Van Raalte, D. H. (2021). Safety of Semaglutide. Frontiers in Endocrinology, 12, 645563. https://doi.org/10.3389/fendo.2021.645563
Wilding, J. P. H., Batterham, R. L., Calanna, S., Davies, M., Van Gaal, L. F., Lingvay, I., McGowan, B. M., Rosenstock, J., Tran, M. T. D., Wadden, T. A., Wharton, S., Yokote, K., Zeuthen, N., & Kushner, R. F. (2021). Once-Weekly semaglutide in Adults with overweight or obesity. New England Journal of Medicine, 384(11), 989–1002. https://doi.org/10.1056/nejmoa2032183
Disclaimer
All products provided by NuScience Peptides are intended for research purposes only. The discussions provided in this blog are purely for education & research purposes. The US FDA (Food & Drug Association) has not approved the drug or any information contained here.
Any potential applications of Semaglutide must be carried out in a controlled research setting, adhering to all relevant ethical and regulatory guidelines. Use of Semaglutide outside of this context is not recommended nor endorsed by NuScience Peptides.
